JT valve works on the principle that when the pressure of a gas/Liquid changes, its temperature also changes.
The Joule–Thomson effect describes the temperature change of a gas or liquid when it is forced through a valve, while kept insulated. ( insulation is required to avoid influence of surrounding environment effects).
JT Valve
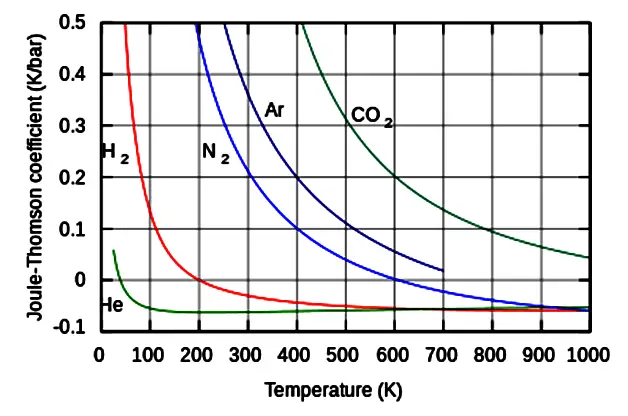
Fig: Joule–Thomson coefficients for various gases at atmospheric pressure.
Joule–Thomson coefficient
The rate of change of temperature (T) with respect to pressure P is a Joule Thomson process (at constant enthalpy).
μJT = (∂T/∂P) H
μJT = Joule Thomson Coefficient expressed in °C/bar or K/Pa
∂T = Change in temperature
∂P = Change in Pressure.
H= Constant Enthalpy.
All real gases have an inversion point at which the μJT changes sign. The temperature of this point the Joule-Thomson inversion temperature, depends on the pressure of the gas before expansion.
First case:
In a gas expansion the pressure decreases below the inversion temperature – the sign ∂P is negative by definition.
With that in mind, if the gas temperature is below the inversion temperature then μJT is positive since ∂P always negative thus ∂T must be negative. So the gas cools.
μJT > 0, then temperature decreases, gas cools.
Second case:
In a gas expansion the pressure decrease and the gas temperature is above the inversion temperature then μJT is negative, ∂P always negative thus ∂T must be positive. So the gas warms.
μJT < 0, then temperature increase, gas warms.
Advantages:
JT valve is used to avoid formation of hydrates down stream of control valve in winter.
Read Next:
- Calculate Control Valve Position
- Sizing the Control Valves
- Valve Relation between Cv and Kv
- Facts About Control Valves
- Control Valve Failure Mode



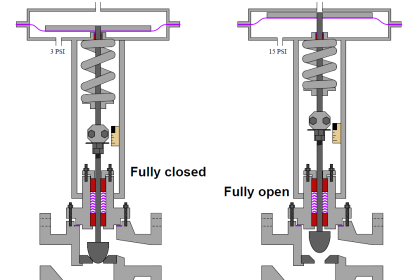





Helpful Points on JT Valve :
1. A normal throttling pressure reducing valve does not have to withstand the stresses and temperature differences imposed on a cryogenic valve;
2. A J-T valve design is used as a by-pass around any cryogenic expansion engine or turboexpander because usually a complete liquefaction of the entire gas stream cannot be allowed to occur within the isentropic expansion device due to the formation of excess harmful/destructive liquid within the machine. Therefore, a J-T valve is applied to a part of the total stream – even though the adiabatic isenthalpic expansion is much less efficient than the isentropic expansion device doing useful work.
3. In carrying out a J-T effect, the expansion valve is subjected to cryogenic temperatures and these require a special design that allows it to function normally when required.
4. An extended stem is a physical requirement for any cryogenic valve in order to insulate the valve body from the required, ambient manual or automatic controls.
5. Due to the cryogenic temperatures, the J-T valve requires exotic materials of construction (stainless steels) that will maintain their tensile strength throughout the cryogenic range.
In Oil or Gas pipe lines, if a Control valve is not operated for prolonged time, in down stream of the valve there are more chances of freezing during winter.
There is a need for more discussion or comments on JT valve with reference to Oi & Gas industry.
how to optimize the recovery when we use the JT valve?