This article provides information on the following methods of producing electricity:
- Electrochemistry
- Static (friction)
- Induction (magnetism)
- Piezoelectric (pressure)
- Thermal (heat)
- Light
- Thermionic emission
Electrochemistry
Chemicals can be combined with certain metals to cause a chemical reaction that will transfer electrons to produce electrical energy. This process works on the electrochemistry principle.
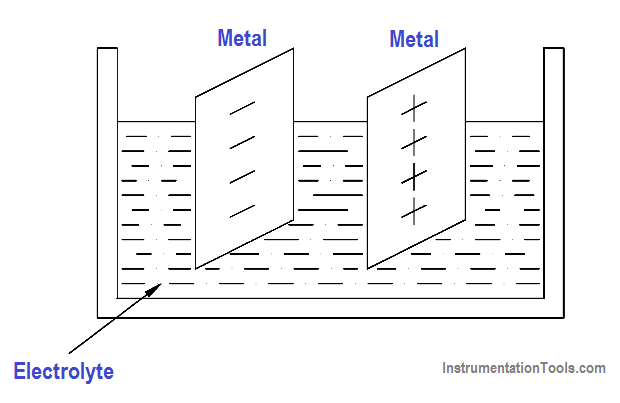
One example of this principle is the voltaic chemical cell, shown in Figure 11. A chemical reaction produces and maintains opposite charges on two dissimilar metals that serve as the positive and negative terminals. The metals are in contact with an electrolyte solution. Connecting together more than one of these cells will produce a battery.
Figure 11 Voltaic Chemical Cell
Example: A battery can maintain a potential difference between its positive and negative terminals by chemical action.
Static Electricity
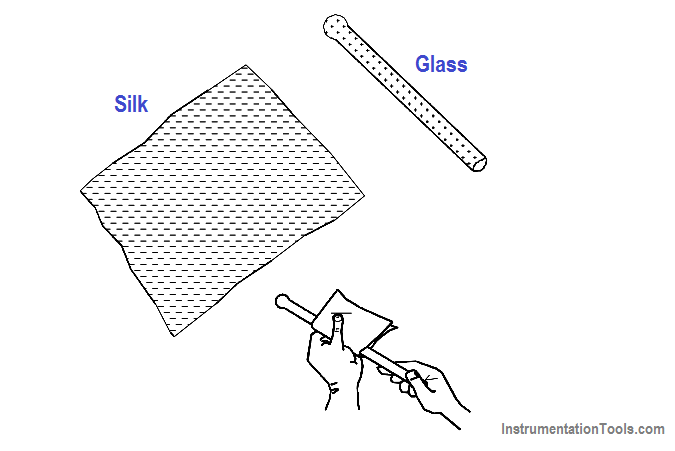
Atoms with the proper number of electrons in orbit around them are in a neutral state, or have a “zero charge.” A body of matter consisting of these atoms will neither attract nor repel other matter that is in its vicinity. If electrons are removed from the atoms in this body of matter, as happens due to friction when one rubs a glass rod with a silk cloth, it will become electrically positive as shown in Figure 12.
If this body of matter (e.g., glass rod) comes near, but not in contact with, another body having a normal charge, an electric force is exerted between them because of their unequal charges. The existence of this force is referred to as static electricity or electrostatic force.
Figure 12 Static Electricity
Example: Have you ever walked across a carpet and received a shock when you touched a metal door knob? Your shoe soles built up a charge by rubbing on the carpet, and this charge was transferred to your body. Your body became positively charged and, when you touched the zero-charged door knob, electrons were transferred to your body until both you and the door knob had equal charges.
Magnetic Induction
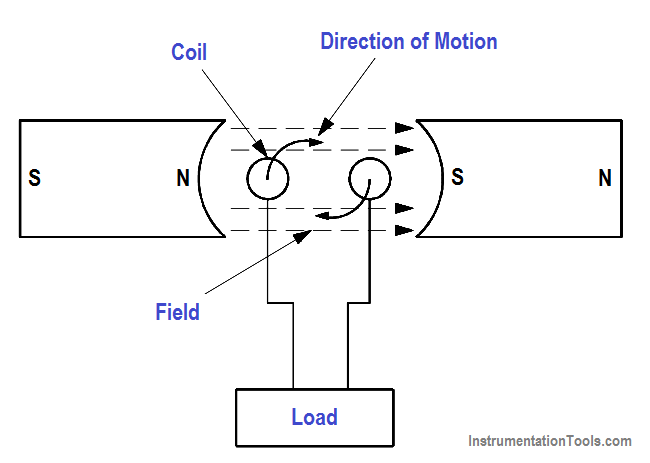
A generator is a machine that converts mechanical energy into electrical energy by using the principle of magnetic induction. Magnetic induction is used to produce a voltage by rotating coils of wire through a stationary magnetic field, as shown in Figure 13, or by rotating a magnetic field through stationary coils of wire. This is one of the most useful and widely employed applications of producing vast quantities of electric power.
Figure 13 – Generator – Electromagnetic Induction
Piezoelectric Effect
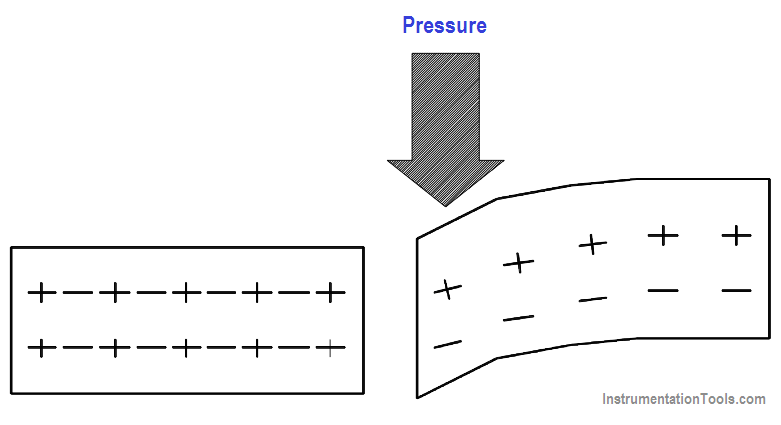
By applying pressure to certain crystals (such as quartz or Rochelle salts) or certain ceramics (like barium titanate), electrons can be driven out of orbit in the direction of the force. Electrons leave one side of the material and accumulate on the other side, building up positive and negative charges on opposite sides, as shown in Figure 14.
When the pressure is released, the electrons return to their orbits. Some materials will react to bending pressure, while others will respond to twisting pressure. This generation of voltage is known as the piezoelectric effect. If external wires are connected while pressure and voltage are present, electrons will flow and current will be produced. If the pressure is held constant, the current will flow until the potential difference is equalized.
When the force is removed, the material is decompressed and immediately causes an electric force in the opposite direction. The power capacity of these materials is extremely small. However, these materials are very useful because of their extreme sensitivity to changes of mechanical force.
Figure 14 Pressure Applied to Certain Crystals Produces an Electric Charge
Example: One example is the crystal phonograph cartridge that contains a Rochelle salt crystal. A phonograph needle is attached to the crystal. As the needle moves in the grooves of a record, it swings from side to side, applying compression and decompression to the crystal. This mechanical motion applied to the crystal generates a voltage signal that is used to reproduce sound.
Thermoelectricity
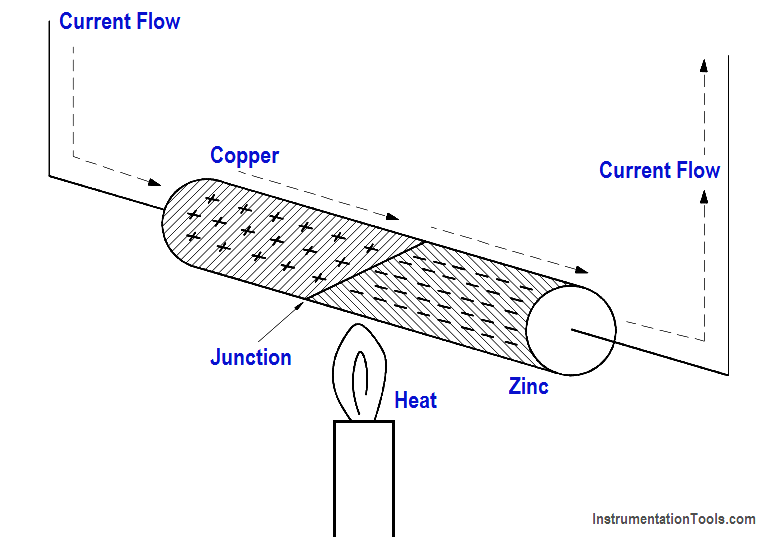
Some materials readily give up their electrons and others readily accept electrons. For example, when two dissimilar metals like copper and zinc are joined together, a transfer of electrons can take place. Electrons will leave the copper atoms and enter the zinc atoms. The zinc gets a surplus of electrons and becomes negatively charged. The copper loses electrons and takes on a positive charge.
This creates a voltage potential across the junction of the two metals. The heat energy of normal room temperature is enough to make them release and gain electrons, causing a measurable voltage potential. As more heat energy is applied to the junction, more electrons are released, and the voltage potential becomes greater, as shown in Figure 15. When heat is removed and the junction cools, the charges will dissipate and the voltage potential will decrease. This process is called thermoelectricity. A device like this is generally referred to as a “thermocouple.”
The thermoelectric voltage in a thermocouple is dependent upon the heat energy applied to the junction of the two dissimilar metals. Thermocouples are widely used to measure temperature and as heat-sensing devices in automatic temperature controlled equipment.
Figure 15 Heat Energy Causes Copper to Give up Electrons to Zinc
Thermocouple power capacities are very small compared to some other sources, but are somewhat greater than those of crystals.
Generally speaking, a thermocouple can be subjected to higher temperatures than ordinary mercury or alcohol thermometers.
Photoelectric Effect
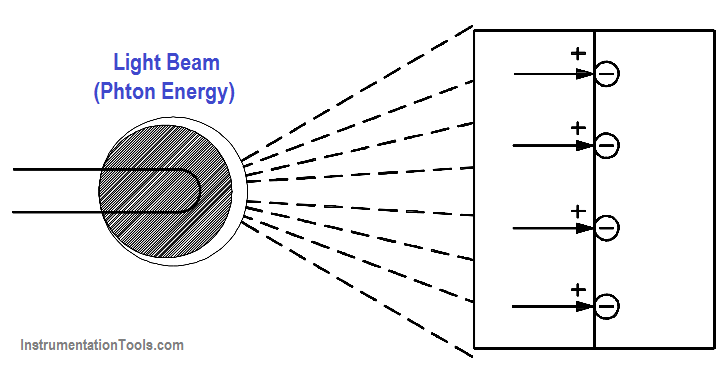
Light is a form of energy and is considered by many scientists to consist of small particles of energy called photons. When the photons in a light beam strike the surface of a material, they release their energy and transfer it to the atomic electrons of the material. This energy transfer may dislodge electrons from their orbits around the surface of the substance. Upon losing electrons, the photosensitive (light sensitive) material becomes positively charged and an electric force is created, as shown in Figure 16.
Figure 16 Producing Electricity from Light Using a Photovoltaic Cell
This phenomenon is called the photoelectric effect and has wide applications in electronics, such as photoelectric cells, photovoltaic cells, optical couplers, and television camera tubes. Three uses of the photoelectric effect are described below.
- Photovoltaic: The light energy in one of two plates that are joined together causes one plate to release electrons to the other. The plates build up opposite charges, like a battery (Figure 16).
- Photoemission: The photon energy from a beam of light could cause a surface to release electrons in a vacuum tube. A plate would then collect the electrons.
- Photoconduction: The light energy applied to some materials that are normally poor conductors causes free electrons to be produced in the materials so that they become better conductors.
Thermionic Emission
A thermionic energy converter is a device consisting of two electrodes placed near one another in a vacuum. One electrode is normally called the cathode, or emitter, and the other is called the anode, or plate. Ordinarily, electrons in the cathode are prevented from escaping from the surface by a potential-energy barrier. When an electron starts to move away from the surface, it induces a corresponding positive charge in the material, which tends to pull it back into the surface.
To escape, the electron must somehow acquire enough energy to overcome this energy barrier. At ordinary temperatures, almost none of the electrons can acquire enough energy to escape. However, when the cathode is very hot, the electron energies are greatly increased by thermal motion. At sufficiently high temperatures, a considerable number of electrons are able to escape. The liberation of electrons from a hot surface is called thermionic emission.
The electrons that have escaped from the hot cathode form a cloud of negative charges near it called a space charge. If the plate is maintained positive with respect to the cathode by a battery, the electrons in the cloud are attracted to it. As long as the potential difference between the electrodes is maintained, there will be a steady current flow from the cathode to the plate.
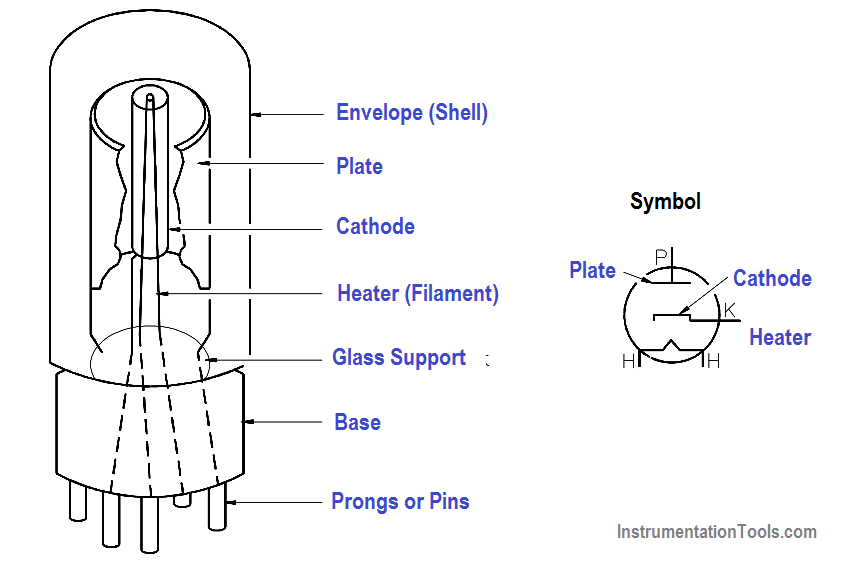
The simplest example of a thermionic device is a vacuum tube diode in which the only electrodes are the cathode and plate, or anode, as shown in Figure 17. The diode can be used to convert alternating current (AC) flow to a pulsating direct current (DC) flow.
Figure 17 Vacuum Tube Diode
Summary
The important information contained in this article is summarized below.
- Electrochemistry – Combining chemicals with certain metals causes a chemical reaction that transfers electrons.
- Static electricity – When an object with a normally neutral charge loses electrons, due to friction, and comes in contact with another object having a normal charge, an electric charge is exerted between the two objects.
- Magnetic induction – Rotating coils of wire through a stationary magnetic field or by rotating a magnetic field through a stationary coil of wire produces a potential.
- Piezoelectric effect – Bending or twisting certain materials will cause electrons to drive out of orbit in the direction of the force. When the force is released, the electrons return to their original orbit.
- Thermoelectricity – Heating two joined dissimilar materials will cause a transfer of electrons between the materials setting up a current flow.
- Photoelectric effect – Dislodging of electrons from their orbits by light beams creates positively-charged objects.
- Thermionic emission – Freeing electrons from a hot surface causes electrons to escape.