Maintaining the calibration of instruments at a large industrial facility is a daunting task. Aside from the actual labor of checking and adjusting calibration, records must be kept not only of instrument performance but also of test conditions and criteria (e.g. calibration tolerance, time interval between calibrations, number of points to check, specific procedures, etc.). A
My practical method to minimize human error in this process is welcome. For this reason, automated and semi-automated calibration tools have been developed to help manage the data associated with calibration, and to make the instrument technician’s job more manageable.
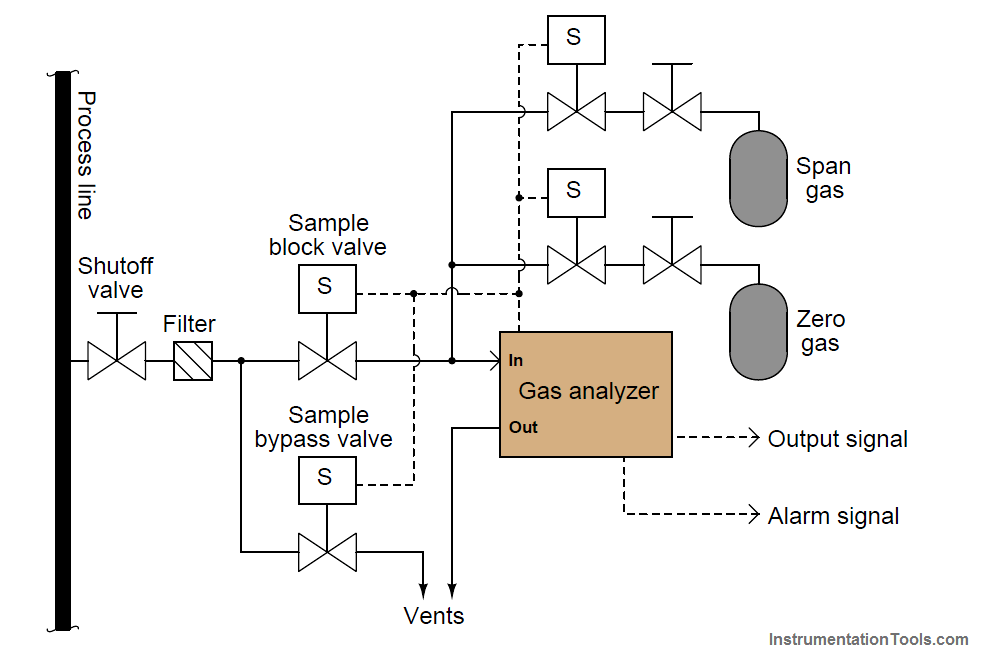
An example of a fully automated calibration system is a process chemical analyzer where a set of solenoid valves direct chemical samples of known composition to the analyzer at programmed time intervals, a computer inside the analyzer recording the analyzer’s error (compared to the known standard) and auto-adjusting the analyzer in order to correct for whatever errors are detected.
In the following illustration we see a schematic of a gas analyzer with two compressed-gas cylinders holding gases of 0% and 100% concentration of the compound(s) of interest, called “zero gas” and “span gas”, connected through solenoid valves so that the chemical analyzer may be automatically tested against these standards:
The only time a human technician need attend to the analyzer is when parameters not monitored by the auto-calibration system must be checked, and when the auto-calibration system detects an error too large to self-correct (thus indicating a fault).
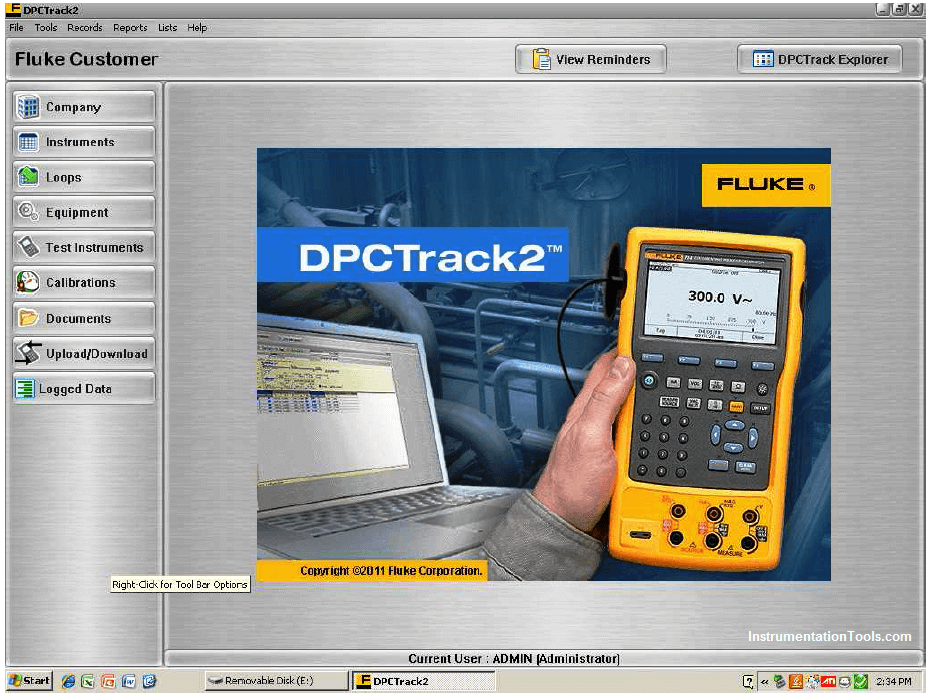
An example of a semi-automated calibration system is an instrument such as Fluke’s series of Documenting Process Calibrators (DPC). These devices function as standards for electrical measurements such as voltage, current, and resistance, with built-in database capability for storing calibration records and test conditions:
A technician using a documenting calibrator such as this is able to log As-Found and As-Left data in the device’s memory and download the calibration results to a computer database at some later time. The calibrator may also be programmed with test conditions for each specific instrument on the technician’s work schedule, eliminating the need for that technician to look up each instrument’s test conditions, and thereby reducing the potential for human error.
An example of database software used to schedule routine instrument calibrations and archive the results is Fluke’s DPCTrack2, a point-and-click user interface serving as a front-end to an SQL database where the instrument data is maintained in digital format on the computer’s hard drive:
Calibration management software allows managers to define calibration schedules, tolerances, and even technician work assignments, the software allowing for downloading of this setup information into a hand-held calibrator unit, as well as uploading and archival of calibration results following the procedure.
In some industries, this degree of rigor in calibration record-keeping is merely helpful; in other industries it is vital for business.
Examples of the latter include pharmaceutical manufacturing, where regulatory agencies (such as the Food and Drug Administration in the United States) enforces rigorous standards for manufacturing quality including requirements for frequent testing and data archival of process instrument accuracy.
Record-keeping in such industries is not limited to As- Found and As-Left calibration results, either; each and every action taken on that instrument by a human being must be recorded and archived so that a complete audit of causes may be conducted should there ever be an incident of product mis-manufacture.