Non-dispersive analyzers employ the principle of spectrographic absorption to measure how much of a particular substance exists within a sample. NDIR gas analyzers shine light through a windowed sample chamber (typically called a cell ), through which a fresh flow of process gas continually moves.
Certain “species” (compounds) of gas within the sample stream absorb part of the incident light, and therefore the light exiting the cell becomes partially depleted of those wavelengths.
A heat sensitive detector placed behind the cell measures how much infrared light did not get absorbed by the sample gas. If we imagine the concentration of light-absorbing gas increasing over time, more of the infrared light entering the cell will being absorbed by the gas and converted into heat within the cell, leaving less light exiting the cell to generate heat at the detector.
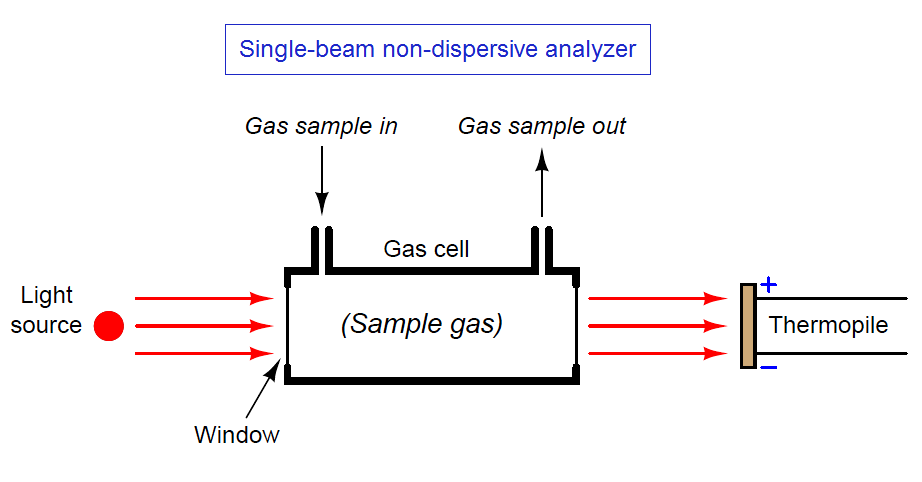
The simplest style of non-dispersive analyzer uses a single light source, shining continuously through a single gas cell, and eventually falling on a small thermopile (converting the received infrared light into heat, and then into a voltage signal):
This crude analyzer suffers from multiple problems. First, it is non-selective: any light-absorbing gas entering the sample cell reduces heat at the detector (i.e. generates less thermopile voltage), regardless of the species. It might work well enough in an application where the only light-absorbing gas in the process mixture happens to be the one gas we are interested in measuring, but most industrial analyzer applications are not like this. In most cases, our process sample contains multiple species of gases capable of absorbing light within a similar range of wavelengths, but we are only interested in measuring one of them.
An example would be the measurement of carbon dioxide (CO2) concentration in the exhaust gas of a combustion furnace: most of the gases exiting the furnace do not absorb infrared light (nitrogen, oxygen), but CO2 gas does. However, carbon monoxide (CO), water vapor (H2O), and sulfur dioxide (SO2) also absorb infrared light, and are all normally present in the exhaust gas of a furnace to varying degrees. Since our crude NDIR analyzer is non-selective, it cannot differentiate between carbon dioxide and any of the other infrared-absorbing gases present in the exhaust gas.
Another significant problem with this analyzer design is that any variations in the light source’s output cause both a zero shift and a span shift in the instrument’s calibration. Since light sources tend to weaken with age, this flaw necessitates frequent re-calibration of the analyzer.
Finally, since the detector is a thermopile, its output will be affected not just by the light falling on it, but also by ambient temperature, causing the analyzer’s output to vary in ways completely unrelated to sample gas composition.