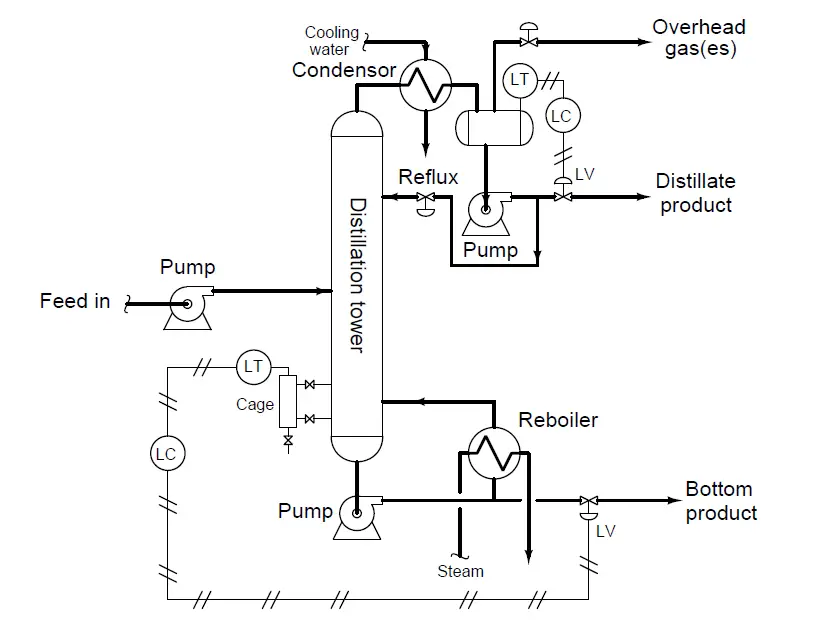
Distillation is a process of continuous boiling and condensing used to separate mixtures of different fluids. A practical example of distillation is the separation of crude oil into multiple gases and liquids (propane, butane, naphtha, hexane, etc.). Components with higher boiling points (typically denser fluids) collect at the bottom of the tower, while components with lower boiling points (typically lighter fluids) and gases collect at the top of the tower.
In this distillation tower, the “feed” fluid is a mixture of just three components, each separated from each other within the distillation tower and draw off as products at three different points. “Overhead” gases are drawn off the very top of the tower, “distillate” liquid is drawn out of an accumulation vessel near the top, and “bottom product” is drawn off the bottom of the tower.
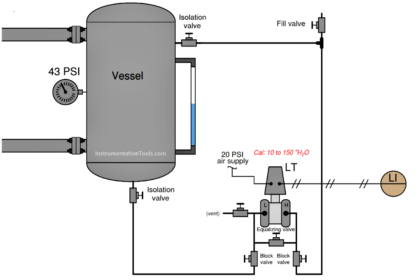
A displacer level transmitter (LT) senses the level of this “bottom product” liquid collecting at the tower’s base:
Displacer Level Instrument
Note: In this example, the level instrument is a pneumatically operated device (old model).
An instrument technician calculates the buoyant force for the displacer of this level instrument to be 0 pounds at 0% liquid level, and 3.75 pounds at 100% liquid level.
Explain how you would check the calibration of this instrument while the distillation tower was running, using these figures.
Hint:
Don’t forget to contact the operator(s) for this process, and either have that operator put the level controller in manual mode, or you do it yourself!
The distillation tower bottom product level will have to be manually controlled for the duration of your calibration test.
More Questions:
- What safety concerns are there in this process that you would have to be aware of?
- What standard will you use for calibration (i.e. the thing you check the instrument’s calibration against)?
- Are there any process-related factors that could skew the calibration accuracy of this instrument while it is in service?
Share your answers with us through below comments section.
Read Next:
- Find LRV and URV
- Strain Gauge Principle
- Solenoid Actuated Valves
- Types of Thermometers
- Basics of Hydrostatic
Credits: Tony R. Kuphaldt