Batteries are used for a wide variety of services throughout technology today. To begin to study battery operation and characteristics, a few terms that are used with batteries must be understood.
They are
- Voltaic cell
- Battery
- Electrode
- Electrolyte
- Specific gravity
- Ampere-Hour
Voltaic Cell
The term voltaic cell is defined as a combination of materials used to convert chemical energy into electrical energy. A voltaic or chemical cell consists of two electrodes made of different types of metals or metallic compounds placed in an electrolyte solution.
Battery
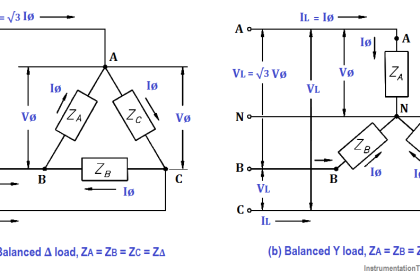
A battery is a group of two or more connected voltaic cells.
Electrode
An electrode is a metallic compound, or metal, which has an abundance of electrons (negative electrode) or an abundance of positive charges (positive electrode).
Electrolyte
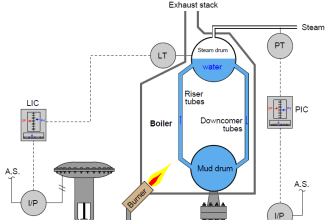
An electrolyte is a solution which is capable of conducting an electric current. The electrolyte of a cell may be a liquid or a paste. If the electrolyte is a paste, the cell is referred to as a dry cell; if the electrolyte is a solution, it is called a wet cell.
Specific Gravity
Specific gravity is defined as the ratio comparing the weight of any liquid to the weight of an equal volume of water. The specific gravity of pure water is 1.000. Lead-acid batteries use an electrolyte which contains sulfuric acid. Pure sulfuric acid has a specific gravity of 1.835, since it weighs 1.835 times as much as pure water per unit volume.
Since the electrolyte of a lead-acid battery consists of a mixture of water and sulfuric acid, the specific gravity of the electrolyte will fall between 1.000 and 1.835. Normally, the electrolyte for a battery is mixed such that the specific gravity is less than 1.350.
Ampere-Hour
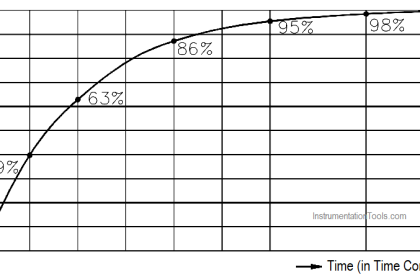
An ampere-hour is defined as a current of one ampere flowing for one hour. If you multiply the current in amperes by the time of flow in hours, the result is the total number of ampere-hours. Ampere- hours are normally used to indicate the amount of energy a storage battery can deliver.
Battery Terminology Summary
- A voltaic cell is a combination of materials used to convert chemical energy into electrical energy.
- A battery is a group of two or more connected voltaic cells.
- An electrode is a metallic compound, or metal, which has an abundance of electrons (negative electrode) or an abundance of positive charges (positive electrode).
- An electrolyte is a solution which is capable of conducting an electric current.
- Specific gravity is defined as the ratio comparing the weight of any liquid to the weight of an equal volume of water.
- An ampere-hour is defined as a current of one ampere flowing for one hour.