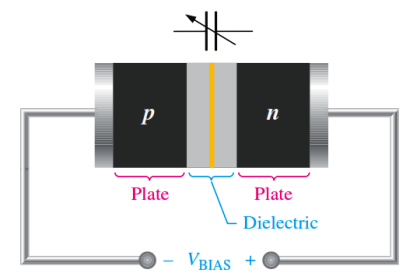
Semiconductive materials do not conduct current well and are of limited value in their intrinsic state. This is because of the limited number of free electrons in the conduction band and holes in the valence band. Intrinsic silicon (or germanium) must be modified by increasing the number of free electrons or holes to increase its conductivity and make it useful in electronic devices. This is done by adding impurities to the intrinsic material. Two types of extrinsic (impure) semiconductive materials, n-type and p-type, are the key building blocks for most types of electronic devices.
Since semiconductors are generally poor conductors, their conductivity can be drastically increased by the controlled addition of impurities to the intrinsic (pure) P. This process, called doping, increases the number of current carriers (electrons or holes). The two categories of impurities are n-type and p-type.
N-Type Semiconductor
To increase the number of conduction-band electrons in intrinsic silicon, pentavalent impurity atoms are added. These are atoms with five valence electrons such as arsenic (As), phosphorus (P), bismuth (Bi), and antimony (Sb).
As illustrated in Below Figure, each pentavalent atom (antimony, in this case) forms covalent bonds with four adjacent silicon atoms. Four of the antimony atom’s valence electrons are used to form the covalent bonds with silicon atoms, leaving one extra electron. This extra electron becomes a conduction electron because it is not involved in bonding. Because the pentavalent atom gives up an electron, it is often called a donor atom. The number of conduction electrons can be carefully controlled by the number of impurity atoms added to the silicon. A conduction electron created by this doping process does not leave a hole in the valence band because it is in excess of the number required to fill the valence band.
Fig: Pentavalent impurity atom in a silicon crystal structure. An antimony (Sb) impurity atom is shown in the center. The extra electron from the Sb atom becomes a free electron.
Majority and Minority Carriers Since most of the current carriers are electrons, silicon (or germanium) doped with pentavalent atoms is an n-type semiconductor (the n stands for the negative charge on an electron). The electrons are called the majority carriers in n-type material. Although the majority of current carriers in n-type material are electrons, there are also a few holes that are created when electron-hole pairs are thermally generated. These holes are not produced by the addition of the pentavalent impurity atoms. Holes in an n-type material are called minority carriers.
P-Type Semiconductor
To increase the number of holes in intrinsic silicon, trivalent impurity atoms are added. These are atoms with three valence electrons such as boron (B), indium (In), and gallium (Ga). As illustrated in Below Figure, each trivalent atom (boron, in this case) forms covalent bonds with four adjacent silicon atoms. All three of the boron atom’s valence electrons are used in the covalent bonds; and, since four electrons are required, a hole results when each trivalent atom is added. Because the trivalent atom can take an electron, it is often referred to as an acceptor atom. The number of holes can be carefully controlled by the number of trivalent impurity atoms added to the silicon. A hole created by this doping process is not accompanied by a conduction (free) electron.
Fig: Trivalent impurity atom in a silicon crystal structure. A boron (B) impurity atom is shown in the center.
Majority and Minority Carriers Since most of the current carriers are holes, silicon (or germanium) doped with trivalent atoms is called a p-type semiconductor. The holes are the majority carriers in p-type material. Although the majority of current carriers in p-type material are holes, there are also a few conduction-band electrons that are created when electron-hole pairs are thermally generated. These conduction-band electrons are not produced by the addition of the trivalent impurity atoms. Conduction-band electrons in p-type material are the minority carriers.