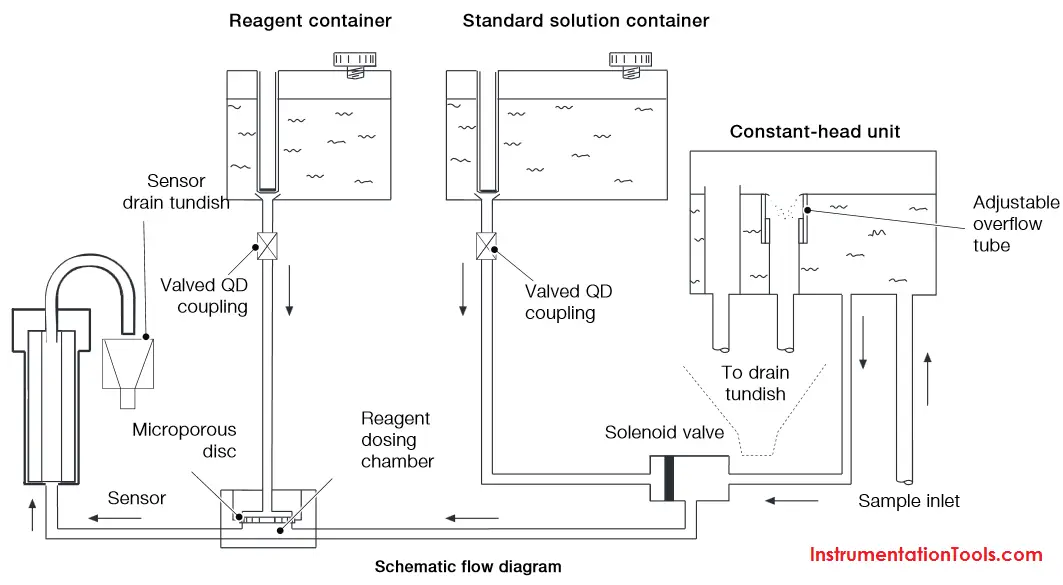
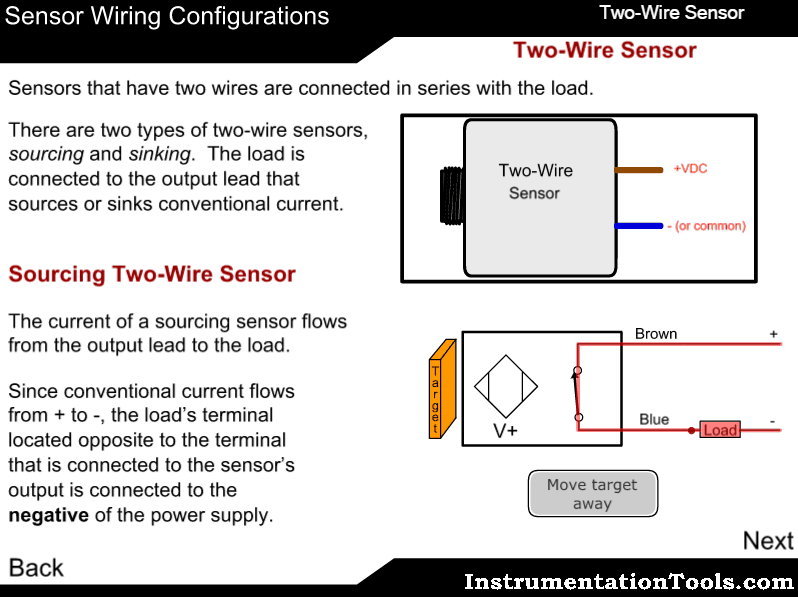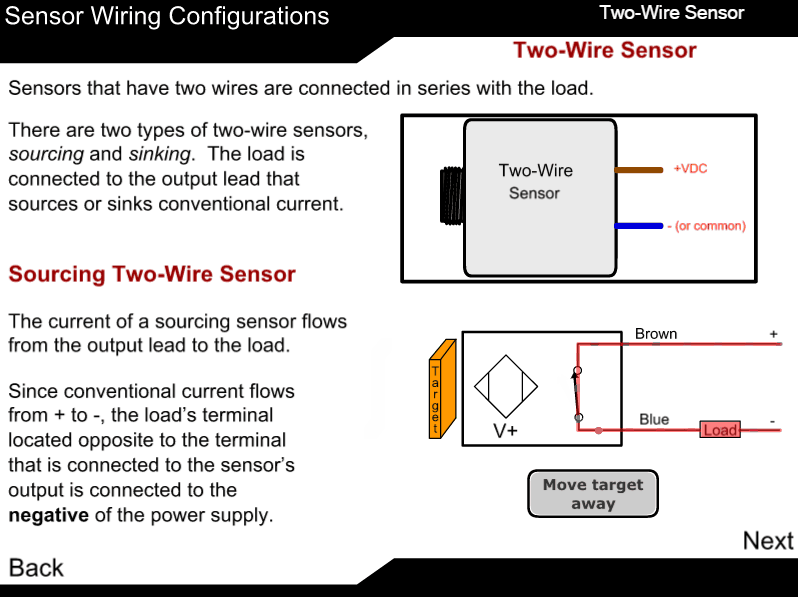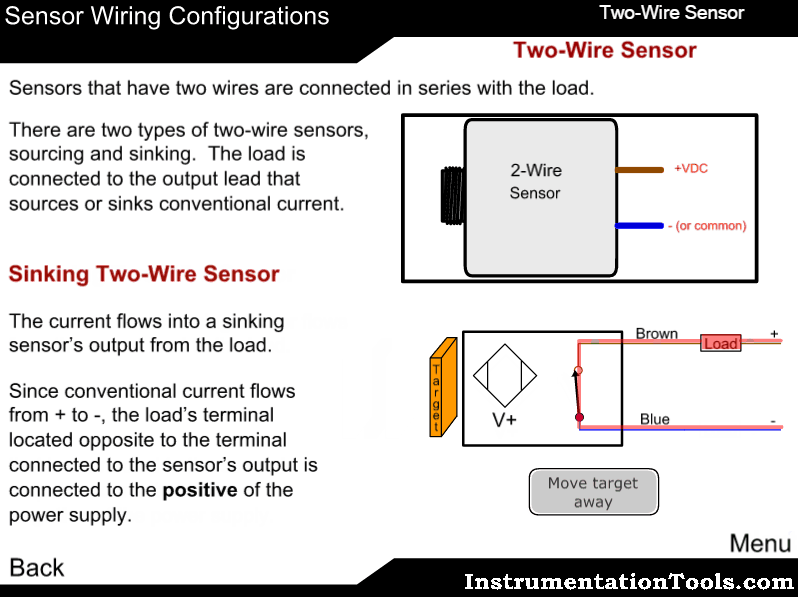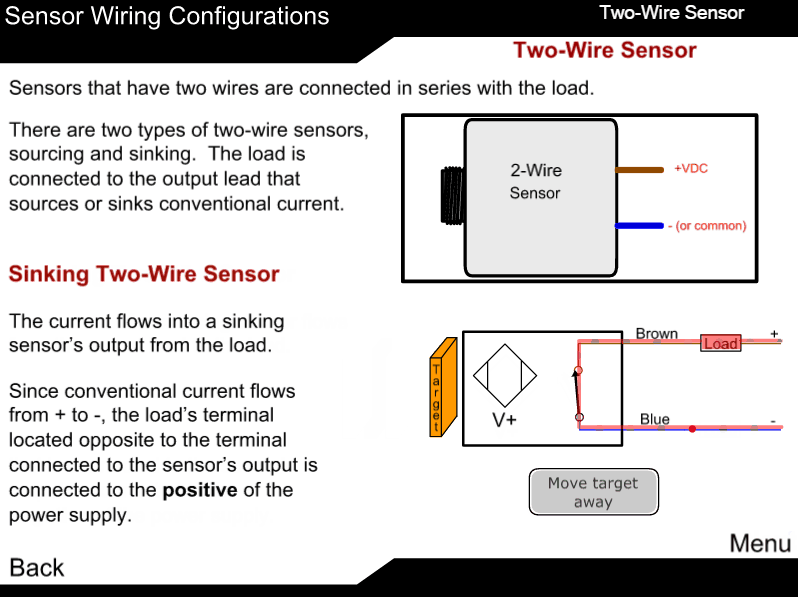
Measuring principle
The sample enters the analyzer via the user-adjustable constant-head unit mounted within the enclosure that removes the effect of changes in sample pressure and flow-rate. Overflow from the constant-head unit drains into a tundish at the bottom of the enclosure.
The constant sample flow passes through the calibration valve manifold into the reagent dosing chamber where an alkali reagent is added via a micro-porous disc to raise the pH of the sample to 10.5. The dosed sample passes through a mixing coil before entering the hydrazine sensor.
The hydrazine sensor comprises a central ceramic tube inside a gel-filled outer jacket. A silver cathode wire is wound round the outer surface of the tube and a spiral platinum anode is inserted down the center. Sample flows up through the tube, over the platinum anode and out to waste. Electrical contact between the two electrodes is made via the ionic transport through the porous ceramic tube.
The resultant current is proportional to the concentration of hydrazine in solution. The hydrazine sensor and its overflow funnel are mounted on a
sub-panel whose height, relative to the standard solution, can be adjusted to provide the correct rate of flow through the sensor.
A temperature sensor, fitted in the hydrazine sensor flowcell, measures the temperature of the sample. The signal from the hydrazine sensor and the temperature sensor is passed to the smart board Controller. The smart board controller accurately calculates the hydrazine measurement result and transfers it digitally to the display.
Applications
Typical applications for the hydrazine analyzer include:
— Monitoring and control of hydrazine dosing of boiler feedwater
— Monitoring dosing efficiency prior to economizer inlet
Hydrazine in boiler feedwater
The need for accurate dosing :
To reduce dissolved oxygenlevels, boiler feedwater is commonly dosed with hydrazine before it enters the boiler. Typically, dosing in a ratio of 3 parts hydrazine to the expected level of dissolved oxygen enables operators to achieve an acceptable concentration of below 5μgkg.
Hydrazine also reacts with soft haematite layers on the boiler tubes, forming a hard magnetite layer that protects the tubes from further corrosion.
To ensure that the correct amount of hydrazine is added to the boiler feedwater, measurements must be taken at both the point of dosing and the economizer feedwater inlet. Adding too little hydrazine results in higher levels of dissolved oxygen in the boiler and impairs the formation of magnetite on the boiler tubes, reducing their resistance to corrosion. Adding too much is unnecessarily wasteful and incurs higher chemical treatment costs.









This words are very useful in my study field