Humidity measurement is an important tool for predicting the climate outdoors as well as controlling the climate indoors. Humidity control is especially important in living, storage, and manufacturing sites.
Definitions
Scientifically stated, humidity is the measure of water vapor present in a gas. Vapor is a term that refers to the gaseous form of a substance that normally exists as a solid or liquid. When liquid exists as a gas, it exerts pressure on its surroundings. This pressure determines the amount of vapor in the air at a given temperature. This value, known as vapor pressure, varies with temperature and pressure, as well as from substance to substance. Water, for example, has a high vapor pressure at temperatures near boiling (at the boiling point the vapor pressure = the atmospheric pressure). A low vapor pressure at temperatures below freezing (all solids generally have low vapor pressures; if they did not, they would evaporate). The vapor pressure rises as pressure decreases, which explains why water boils at a lower temperature in higher altitudes (low pressure). Absolute humidity is a measure of the mass of the water vapor present in a specified volume. Because the mass of water vapor is difficult to measure, a more common measurement called relative humidity is used. Relative humidity (RH) is the percentage of the amount of water that the air can hold at a given temperature. The following equation calculates the percent relative humidity.
Where:
Pa = actual pressure
Ps = saturated pressure
Relative Humidity is temperature dependent. At 100% RH, the actual pressure of the water vapor is equal to the saturation pressure. The temperature where this exists is called the dew point. Any cooling below the dew point causes water to condense. If the atmosphere stabilizes at a constant humidity during the day, the falling temperatures at night may go below the dew point causing the vapor to condense. The condensing vapor creates the phenomenon commonly known as dew. A crucial fact to note is that it is only necessary for the sensor to obtain one measurement, absolute humidity, RH, or dew point, because the other two can be calculated using the ambient temperature, graphs or equations.
Sensing Methods
In general, obtaining a measure of humidity is not an easy task. Many of the instruments have poor accuracy, narrow bandwidth, contamination problems, and hysteresis. Some of the instruments are large, awkward, and expensive pieces of equipment. To compound the problem there are few highly accurate devices for humidity generation and measurement, needed for use in sensor calibration. Discussed below, are the different methods of humidity sensing.
Psychrometric Method
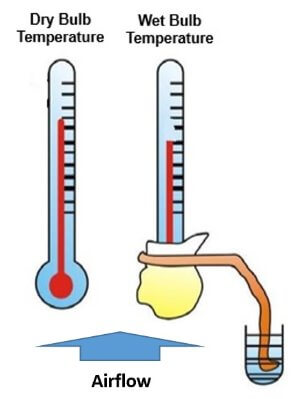
The oldest method for measuring relative humidity is the psychrometric method. Psychrometry is commonly known as the “wet” and “dry” bulb method. A psychrometric sensor does not directly sense humidity, but rather it senses temperature to indirectly find relative humidity. The sensing elements can be thermometers, RTD’s, or thermistors. The first sensing element, the dry bulb, measures ambient temperature. The second sensing element, the wet bulb, is enclosed in a wick saturated with distilled water. Air forced across the wet bulb creates evaporation, which cools it below ambient temperature. The amount of evaporation (cooling) is dependent on the vapor pressure of the air. Using the wet and dry bulb temperatures, the relative humidity can be looked up on a psychrometric chart. Looking up the %RH on a chart for every measurement is time-consuming and cumbersome. With today’s technology, psychrometric charts and dew point equations can be stored in a microprocessor, thus making this a direct sensing method for RH and dew point.
The sling psychrometer dates back to the late 19th century. It used mercury thermometers for temperature measurement, and swinging the bulb around created air movement across the wet bulb. Nowadays, the units have built in fans to ventilate the wet bulb. A psychrometric sensor has good precision with %RH resolutions of 0.1%, humidity ranges from 10 – 100% at temperatures from 32oF to 140oF, and accuracy’s of + 2%. The drawbacks of a psychrometric sensor are a slow response time and they are substantially more expensive.
Dew Point Method
Another method for measuring humidity is using a dew point sensor. There are two common types of dew point sensors, the cooled condensation surface type, or a solution of saturated lithium chloride. The solution of saturated lithium chloride does not directly sense relative humidity. Saturating a wick with resistive electrodes in the solution and an excitation current through the wick creates joule heating. The heating causes some of the solution to evaporate, which reduces the resistance and slows heating. Eventually equilibrium is reached and the temperature of the solution can then be related to the dew point.
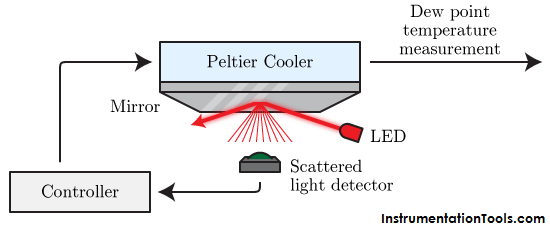
The cooled condensation surface type detects very accurately the temperature at which condensation begins. The most common device used is a mirror to detect for condensation. The system is set up so an LED (light emitting diode) reflects off the mirror at an angle of about 45 degrees. A photo-transistor detects the reflected light. Then, the temperature of the mirror is electronically controlled. The system works by cooling the mirror’s surface below the ambient temperature until condensation forms. The condensation on the surface of the mirror causes the LED’s light to scatter. The scattered light creates a sudden drop in the output of the photo-transistor. At this point, the surface temperature of the mirror is read using a temperature sensor such as an RTD or thermistor. This temperature is the dew point. With a feedback loop, the cooling or heating of the mirror continuously tracks the dew point. There are a few different designs of condensation surfaces that are also used. The chilled mirror method is the most stable and accurate method to determine relative humidity. It is crucial to keep the mirror clean and ensure the temperature sensor and mirror is of high quality. This method has the best humidity range (0-100%RH) and can be used for numerous gases at many pressures. These instruments are bulky and very expensive.
Hygrometric Method
The hygrometric method of relative humidity sensing is the most common. The instruments are generally compact, reliable, and inexpensive. Hygrometric humidity sensors provide an output that is directly indicative of humidity. The first humidity sensing elements were mechanical in nature. Physical dimensions of various materials will change with the adsorption1 of water. Some examples of these are hair, animal membrane, and some plastics. To build a sensor from these materials the element is kept in tension with a spring. A strain gage monitors the displacement caused by a change in the moisture content of the air. The output of the strain gage is directly proportional to the relative humidity. A second method of hygrometry is coating an oscillating crystal (quartz) with a hygroscopic coating. When the coating adsorbs water the mass changes which then changes the crystal’s oscillating frequency. A more obscure method is an electrolytic hygrometer. This method is complicated and not used frequently enough to warrant explanation.
Recent strides in thin film and micromachining technology make it possible to produce high quality resistive and capacitive hygrometric sensors. These sensors have become more accurate, compact, and stable in recent years making them popular in industry. The materials used to produce these sensing elements have the ability to change their electrical characteristics with the adsorption of water. The materials have changed over the years from electrolytic salts, to ceramics, to the recently popular polymers. The new designs in sensing material have overcome many problems. The first major problem with the sensors was narrow bandwidths. Each individual sensor was only reliable over a span of 10 to 20% relative humidity. Numerous manufactured sensors with specific spans cover the entire %RH span. Then there was a problem when adsorbed water left impurities behind on the surface. These impurities would change the electrical characteristics of the sensing materials. The polymers used now have overcome these early problems.
A capacitive sensor is built like a parallel-plate capacitor. The sensing element serves as the dielectric. As the moisture in the air changes the water vapor, the sensing polymer changes with adsorption, resulting in a dielectric constant change. The dielectric constant is directly proportional to the capacitance, which is inversely proportional to the %RH. New techniques in producing thin films have made these, accurate, stable, and easy to manufacture large quantities.
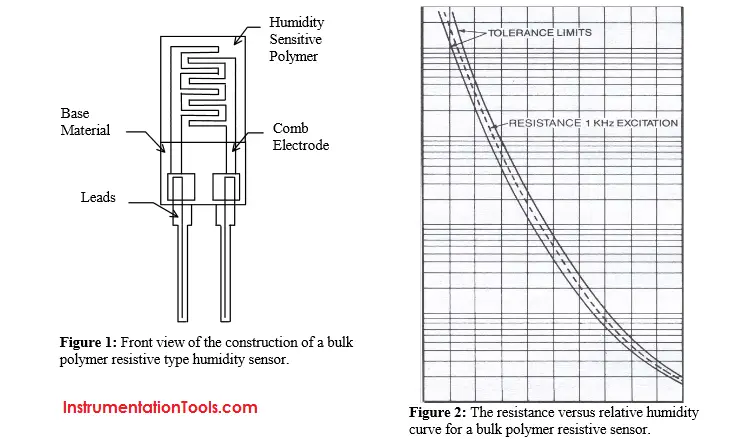
The resistive types of hygrometers are the sensors ACI uses in their products. The sensors consist of placing a thin film of sensing polymer over a set of comb electrodes. Figure 1 is a representation of the physical parts of the humidity sensor. The sensor adsorbs water into the humidity sensing material, which changes the polymers resistance. Other materials adsorb the water, which changes just the surface resistivity. Because the water is absorbed the bulk resistance of the polymer changes making the sensor resistant to surface contamination. The sensor requires an AC excitation current. So there is no chance electrolysis or separation of the humidity-sensing polymer can occur. Thin film technology makes these sensors accurate, stable, and easy to manufacture. The choice of material assures they have fast response times with little hysteresis. Another attraction is their small size and low cost.
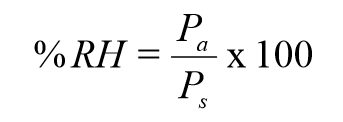







nice reading and learning material
Congratulations Mr. Bharadwaj Reddy for all the wonderful information that you keep sharing through this portal. I find it very useful for my teaching profession to Instrumentations students.
Keep up the good work!
For capacitive sensor : If there is different in comparing with salt saturation method, how to calibrate :
1)Possible to calibrate the sensor ?
2) Using correction factor ?