Question 1:
Water, at its maximum density (temperature of 3.98o C, or approximately 4o C), weighs 62.428 lb/ft3. Based on this reference density, determine the specific gravities of the following liquids:
Sea water: D = 63.99 lb/ft3
Mercury: D = 849 lb/ft3
Olive oil: D = 57.3 lb/ft3
Castor oil: D = 60.5 lb/ft3
Kerosene: D = 51.2 lb/ft3
Benzene: D = 56.1 lb/ft3
Acetone: D = 49.4 lb/ft3
Glycerin: D = 78.6 lb/ft3
Methanol (methyl alcohol): D = 49.41 lb/ft3
Ethanol (ethyl alcohol): D = 49.27 lb/ft3
Ethylene glycol (ethanediol): D = 69.22 lb/ft3
Gasoline: D = 41 lb/ft3 to 43 lb/ft3
Answer:
Sea water: 1.025
Mercury: 13.6
Olive oil: 0.918
Castor oil: 0.969
Kerosene: 0.820
Benzene: 0.899
Acetone: 0.791
Glycerin: 1.26
Methanol: 0.7915
Ethanol: 0.7892
Ethylene glycol: 1.109
Gasoline: 0.657 to 0.689
Question 2:
Toluene has a density of 0.8669 g/cm3 at 20o C. Calculate its density in units of pounds per cubic feet and its specific gravity (unitless).
Answer:
Specific gravity = 0.8669 (same as density in units of g/cm3 )
The units of grams and cubic centimeters are defined in such a way that their density quotient in relation to pure water is 1. This is similar to the Celsius temperature scale, similarly defined at the 0o and 100o points by the freezing and boiling points of pure water, respectively.
To calculate the density of toluene in units of pounds per cubic feet, simply multiply the density of water (62.428 lb/ft3 ) by the specific gravity of toluene (0.8669):
(62.428 lb/ft3 )(0.8669) = 54.12 lb/ft3
Read Next:
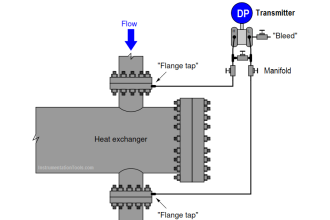
- Pneumatic DP Transmitter
- Calculate Hydrostatic Pressure
- Block and Bleed Valves
- Air Compressor Problem
- PID Control Loop Faults
Credits: Tony R. Kuphaldt