Categories of Gas Monitoring :
1. Combustible/ Flammable Gas
• Explosive hazard.
• To avoid an explosion, atmospheric levels must be maintained below the lower explosive limit (LEL) for each gas, or purged of oxygen.
• Generally measured as 0-100% of the lower explosive limit or in part per million range.
• Combustible gas monitors are designed to alarm before a potential explosive condition occurs.
2. Toxic/ Irritant Gases
• Hazardous to human health; worker exposure must be monitored.
• Typically measured in the part per million (ppm) range.
• Toxic gas monitors are designed to alert workers before the gas level reaches a harmful concentration.
• Some toxic gas monitors can calculate the average exposure over time, providing short-term exposure limit (STEL) and time-weighted average (TWA) readings.
3. Oxygen
• Atmospheres containing too little oxygen (less than 19.5% oxygen by volume) are considered “oxygen deficient” and interfere with normal human respiration.
• Atmospheres containing too much oxygen (more than 25% oxygen by volume) are considered “oxygen enriched” and possess an increased risk of explosion.
• Measured in the percent volume range (normal oxygen percentage in air is 20.8% by volume at sea level).
• Oxygen monitors are generally set to alarm if the atmosphere contains either too little or too much oxygen.
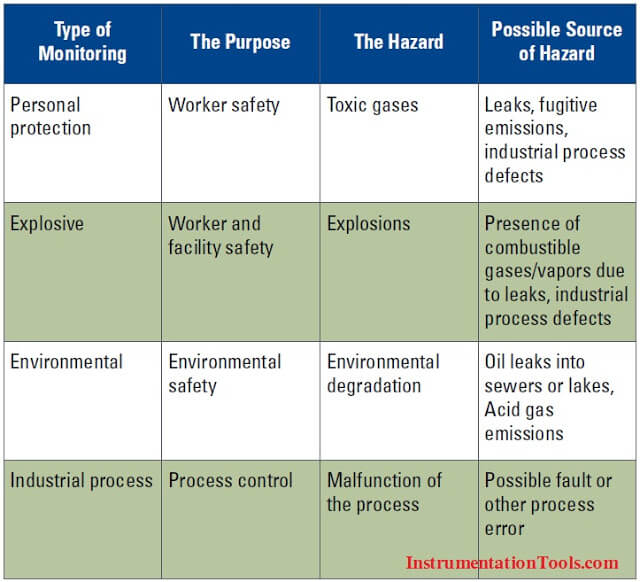
The Four Main Types of Gas Hazards
The following table summarizes the four main reasons why gas monitoring is performed:
Combustible Atmospheres
In order for a flame to exist, three conditions must be met. There must be:
• A source of fuel (e.g. methane or gasoline vapors).
• Enough oxygen (greater than 10-15%) to oxidize or burn the fuel.
• A source of heat (ignition) to start the process.

Examples of Heat and Ignition Sources
• Open flames such as those from lighters, burners, matches and welding torches are the most common sources of ignition.
• Radiation in the form of sunlight or coming from hot surfaces.
• Sparks from various sources such as the switching on or off of electric appliances, removing plugs, static electricity or switching relays.
Combustible Atmosphere Factors
Vapor vs. Gas
Though often used interchangeably, the terms “vapor” and “gas” are not identical. The term “vapor” is used to refer to a substance that, though present in the gaseous phase, generally exists as a liquid or solid at room temperature. When we say that a liquid or solid substance is burning, it is actually its vapors that burn. “Gas” refers to a substance that generally exists in the gaseous phase at room temperature.
Vapor Pressure and Boiling Point
Vapor pressure is the pressure exerted when a solid or liquid is in equilibrium with its own vapor. It is directly related to temperature. An example of vapor pressure is the pressure developed by the vapor of a liquid in a partially-filled closed container. Depending on temperature, the vapor pressure will increase up to a certain threshold. When this threshold is reached, the space is considered to be saturated.
The vapor pressure and boiling point of a chemical determine how much of it is likely to become airborne. Low vapor pressure means there are less molecules of the substance to ignite, so there is generally less of a hazard
present. This also means that there are less molecules to sense, which may make detection more challenging and require higher-sensitivity instrumentation. With higher vapor pressure and a lower boiling point, there is a greater likelihood of evaporation. If containers of chemicals with such properties are left open, or if they’re allowed to spread over large surfaces, they are likely to cause greater hazards.
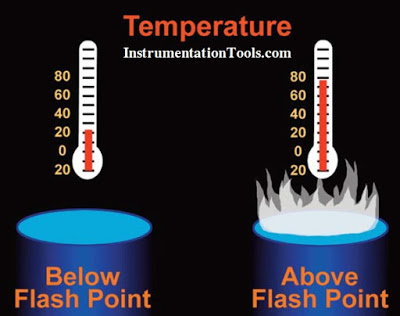
Flashpoint
A flammable material will not give off an amount of gas or vapor sufficient to start a fire until it is heated to its flashpoint. Flashpoint is defined as the lowest temperature at which a liquid produces sufficient vapor to produce a flame. If the temperature is below this point, the liquid will not produce enough vapor to ignite. If the flashpoint is reached and an external source of ignition such as a spark is provided, the material will catch fire. The National Fire Protection Agency’s NFPA’s document NFPA-325M, Fire Hazard Properties of Flammable Liquids, Gases and Volatile Solvents, lists the flashpoints of many common substances.
Flash points are significant because they give an indication of the degree of hazard presented by a flammable liquid. Generally, the lower the flash point, the easier it is for flammable fuel-air mixtures to form, and thus the greater hazard.
Autoignition Temperature
If heated to a certain point—the spontaneous ignition (or “autoignition”) temperature—most flammable chemicals can spontaneously ignite under its own heat energy, without an external source of ignition.
Vapor Density
Vapor density is the weight ratio of a volume of flammable vapor compared to an equal volume of air. Most flammable vapors are heavier than air so they gravitate toward the ground, settling in low areas. A gas or vapor with a vapor density greater than 1 may travel at low levels to find a source of ignition (e.g. hexane, which has a 3.0 vapor density); a gas or vapor with a vapor density less than 1 will tend to rise (e.g. methane, which has a 0.6 vapor density). Vapor density is important to consider when determining optimum sensor placement because it helps predict where the gas or vapor is most likely to accumulate in a room or area.
Explosive Limits
To produce a flame, a sufficient amount of gas or vapor must exist. But too much gas can displace the oxygen in an area and fail to support combustion. Because of this, there are limits at both low-end and high-end gas concen- trations where combustion can occur. These limits are known as the Lower Explosive Limit (LEL) and the Upper Explosive Limit (UEL). They are also referred to as the Lower Flammability Limit (LFL) and the Upper Flammability Limit (UFL).
To sustain combustion, the atmosphere must contain the correct mix of fuel and oxygen (air). The LEL indicates the lowest quantity of gas which must be present for combustion and the UEL indicates the maximum quantity of gas. The actual LEL level for different gases may vary widely and are measured as a percent by volume in air. Gas LELs and UELs can be found in NFPA 325.
LELs are typically 1.4% to 5% by volume. As temperature increases, less energy is required to ignite a fire and the percent gas by volume required to reach 100% LEL decreases, increasing the hazard. An environment containing enriched oxygen levels raises the UEL of a gas, as well as its rate and intensity of propagation. Since mixtures of multiple gases add complexity, their exact LEL must be determined by testing.
Most combustible gas instruments measure in the LEL range and display gas readings as a percentage of the LEL. For example: a 50% LEL reading means the sampled gas mixture contains one-half of the amount of gas necessary to support combustion.

Any gas or vapor concentration that falls between these two limits is in the flammable (explosive) range. Different substances have different flammable range widths — some are very wide and some are narrower. Those with a wider range are generally more hazardous since a larger amount of concentration levels can be ignited.

Atmospheres in which the gas concentration level is below the LEL (insufficient fuel to ignite) are referred to as too “lean” to burn; those in which the gas level is above the UEL (insufficient oxygen to ignite) are too “rich” to burn.
Toxic Atmospheres
Toxic Gas Monitoring
A toxic gas is one which is capable of causing damage to living tissue, impairment of the central nervous system, severe illness or—in extreme cases—death, when ingested, inhaled or absorbed by the skin or eyes. The amounts required to produce these results vary widely with the nature of the substance and exposure time. “Acute” toxicity refers to exposure of short duration, such as a single brief exposure. “Chronic” toxicity refers to exposure of long duration, such as repeated or prolonged exposures.
Toxic gas monitoring is important because some substances can’t be seen or smelled and have no immediate effects. Thus the recognition of a gas hazard via a worker’s senses often comes too late, after concentrations have reached harmful levels.
The toxic effects of gases range from generally harmless to highly toxic. Some are life-threatening at even short, low-level exposures, while others are hazardous only upon multiple exposures at higher concentrations. The degree of hazard that a substance poses to a worker depends upon several factors which include the gas concentration level and the duration of exposure.
Exposure Limits
The American Conference of Governmental Industrial Hygienists (ACGIH) publishes an annually revised list of recommended exposure limits for common industrial compounds, titled “TLV“s and BEI“s Based on the Documentation of the Threshold Limit Values for Chemical Substances and Physical Agents and Biological Exposure Indices”. (To order a copy, see www.acgih.org). ACGIH developed the concept of Threshold Limit Value“ (TLV), which is defined as the airborne concentration of a contaminant to which it is believed that almost all workers may be repeatedly exposed, day after day, over a working lifetime without developing adverse effects. These values are based on a combination of industrial experience and human and animal research.
Time Weighted Averages (TWAs)
TLVs are generally formulated as 8-hour time-weighted averages. The averaging aspect enables excursions above the prescribed limit as long as they are offset by periods of exposure below the TLV.
Short-Term Exposure Limits (STELs)
Short-term exposure limits are concentrations above the 8-hour average to which workers may be exposed for short periods of time without harmful effects. (If the concentration is high enough, even a one-time exposure can produce harmful health effects.) STELs are used to govern situations in which a worker is exposed to a high gas concentration, but only for a short period of time. They are defined as 15-minute time-weighted averages that are not to be exceeded even if the 8-hour TWA is below the TLV.
Ceiling Concentrations
For some toxic gases, a single exposure exceeding the TLV may be hazardous to worker health. In these cases, ceiling concentrations are used to indicate levels that are never to be exceeded.
Permissible Exposure Limits (PELs)
PELs are enforced by the Occupational Safety and Health Administration (OSHA). Part 29 of the Code of Federal Regulations (CFR) Section 1910.1000 contains these standards, which are similar to ACGIH TLVs except that they are legally enforceable rather than simply recommendations. However, the most accurate PELs are listed in the associated Material Safety Data Sheets (MSDS).
Immediately Dangerous to Life and Health (IDLH)
The National Institute for Occupational Safety and Health (NIOSH) defines an IDLH exposure condition atmosphere as one that poses a threat of exposure to airborne contaminants when that exposure is likely to cause death or immediate or delayed permanent adverse health effects or prevent escape from such an environment. Since IDLH values exist to ensure that a worker can escape from a hazardous environment in the event of failure of respiratory protection equipment, they are primarily used to determine appropriate respiratory selection in compliance with OSHA standards.
Gas detection systems are used to monitor toxic gases in primarily two types of monitoring applications:
1. Ambient air monitoring (includes leak monitoring)
• low-level gas detection for worker safety
• to reduce leakage of expensive compounds (e.g., refrigerants)
2. Process monitoring
• to monitor levels of compounds used in chemical synthesis processes (e.g., in the plastics, rubber, leather and food industries)
• from low ppm levels to high % by volume levels
For toxic gas monitoring, electrochemical, metal oxide semiconductor (solid state), infrared and photoionization are the sensing technologies most commonly used.
Oxygen Deficiency/Enrichment
Oxygen Deficiency
Normal ambient air contains an oxygen concentration of 20.8% by volume. When the oxygen level dips below 19.5% of the total atmosphere, the area is considered oxygen deficient. In oxygen-deficient atmospheres, life-supporting oxygen may be displaced by other gases, such as carbon dioxide. This results in an atmosphere that can be dangerous or fatal when inhaled. Oxygen deficiency may also be caused by rust, corrosion, fermentation or other forms of oxidation that consume oxygen. As materials decompose, oxygen is drawn from the atmosphere to fuel the oxidation process.
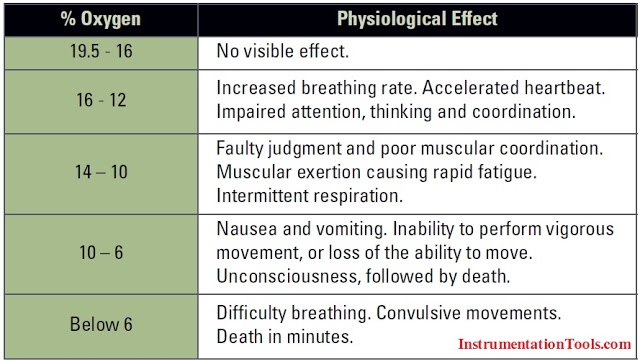
The impact of oxygen deficiency can be gradual or sudden, depending on the overall oxygen concentration and the concentration levels of other gases in the atmosphere. Typically, decreasing levels of atmospheric oxygen cause the following physiological symptoms:
Oxygen Enrichment
When the oxygen concentration rises above 20.8% by volume, the atmosphere is considered oxygen-enriched and is prone to becoming unstable. As a result of the higher oxygen level, the likelihood and severity of a flash fire or explosion is significantly increased.













Good job sir,
Waiting here to learn more topics about instrumentation
Also update regarding loop diagrams, Panel wiring
thanks very good
This is very good web site lot of knowledge about instrument gained
Thanks